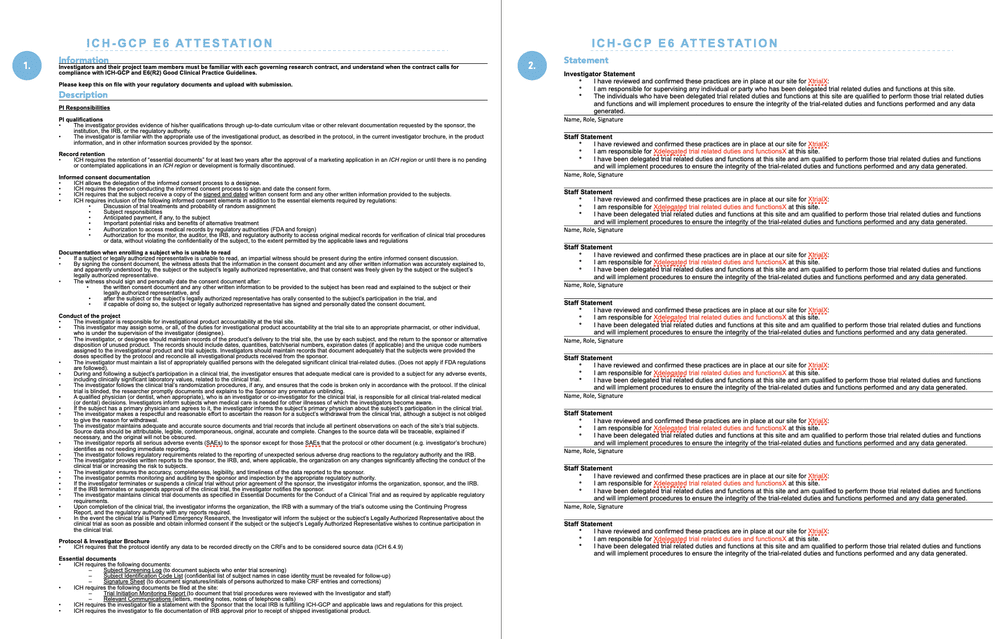
CAPA and Root Cause Analysis: Essentials Post ICH GCP E6 (R2) Addendum - Life Science Training Institute

REFRESHER: ICH Good Clinical Practice (GCP) E6 (R2) and regulatory requirements for Clinical Trials (Fiona Stanley Hospital) - RETProgram
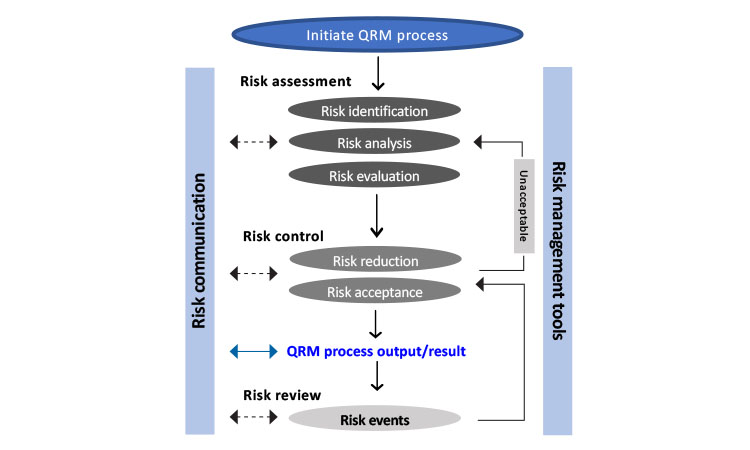
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering
1.) Go to Website: GLOBAL HEALTH TRAINING CENTRE a. https://globalhealthtrainingcentre.tghn.org/ b. REGISTER ( TOP RIGHT CORNER)

Final ICH GCP E6 R2 Addendum: Overview of Changes Impacting Sponsors/CROs/ Clinical Investigator/Site - YouTube

The Impact of ICH GCP E6 Guideline R2 Revisions on Sponsors, Sites, Contract Research Organizations and Vendors | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

E6(R2) Good Clinical Practice Integrated Addendum to ICH E6(R1) - E6(R2) Good Clinical Practice: - Studocu

.jpg)