
Early phase clinical trials extension to guidelines for the content of statistical analysis plans | The BMJ

The GetReal Trial Tool: design, assess and discuss clinical drug trials in light of Real World Evidence generation - Journal of Clinical Epidemiology

Methodological and Statistical Considerations in Clinical Research Dr. Gloria Crispino, CStat CMath Copy Rights; Do not reproduce without authorization. - ppt download

PPT - Robert O'Neill , Ph.D. Director, Office of Biostatistics, CDER, FDA PowerPoint Presentation - ID:7035315

ICH E9 guideline 'Statistical principles for clinical trials': a case study Response to A. Phillips and V. Haudiquet - Brown - 2003 - Statistics in Medicine - Wiley Online Library

Book 13: 2021 FDA Guidance on Clinical Study Reports and Statistical P – Clinical Research Resources, LLC
1097-0258(19990815)18:15%3C1903::aid-sim188%3E3.0.co;2-f.fp.png)
Statistical principles for clinical trials (ICH E9): an introductory note on an international guideline - Lewis - 1999 - Statistics in Medicine - Wiley Online Library

Session 4: Analysis and reporting Managing missing data Rob Coe (CEM, Durham) Developing a statistical analysis plan Hannah Buckley (York Trials Unit) - ppt download
![Seven steps for statistical success in clinical trials [Good clinical practice guidelines] Part 1 – ACTA – Australian Clinical Trials Alliance Seven steps for statistical success in clinical trials [Good clinical practice guidelines] Part 1 – ACTA – Australian Clinical Trials Alliance](https://clinicaltrialsalliance.org.au/wp-content/uploads/2020/10/James-Matcham-Photo_2020-500x334.jpg)









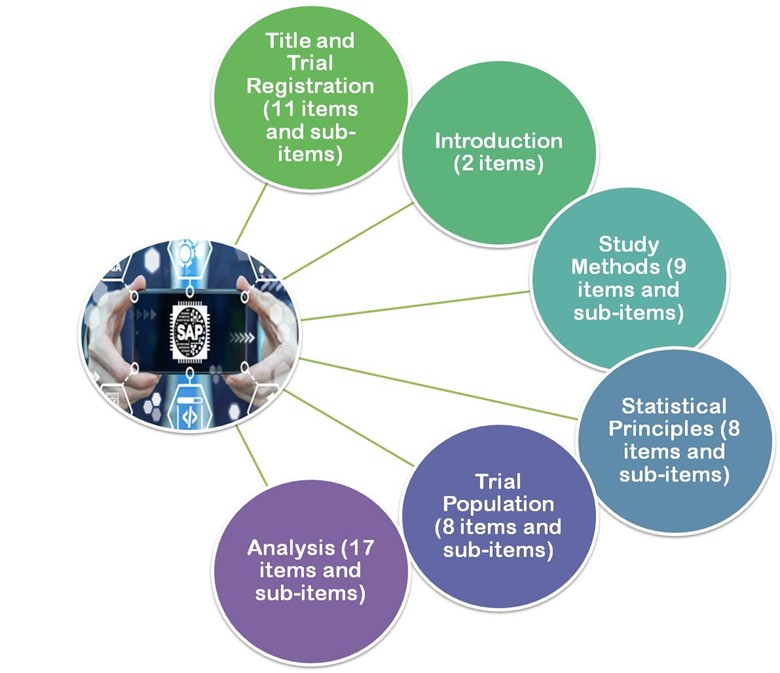

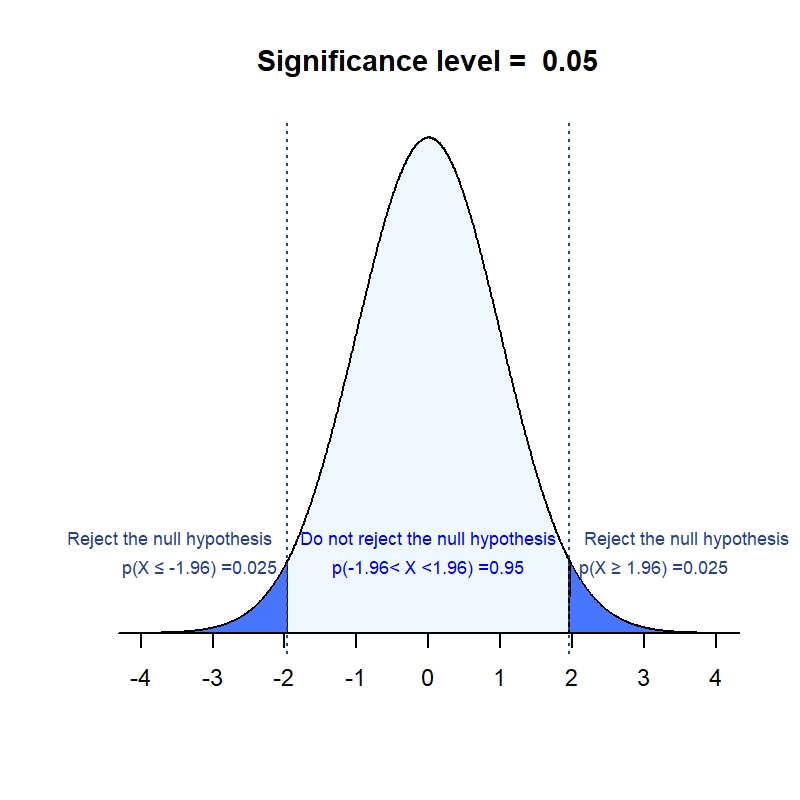

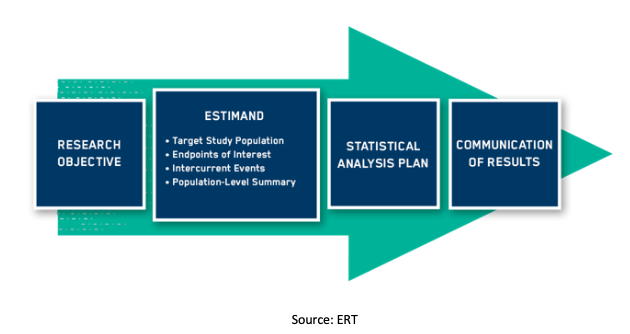


![Understanding addendum to Statistical Principles for Clinical Trials [ICH E9 (R1)] Understanding addendum to Statistical Principles for Clinical Trials [ICH E9 (R1)]](https://media-exp1.licdn.com/dms/image/C5612AQHs6JfqjIC0lw/article-cover_image-shrink_600_2000/0/1520167027524?e=1648684800&v=beta&t=4FAQjVfZN_XdLJjgH966wkAC92tvEfwlnURv-knA55U)

