Guide to Clinical Trials Authorised for Conduct under the Clinical Trials Directive (Council Directive 2001/20/EC) in Ireland

Book 6: 2023 Clinical Trials in The EU: Selected Legislation, Guidelin – Clinical Research Resources, LLC

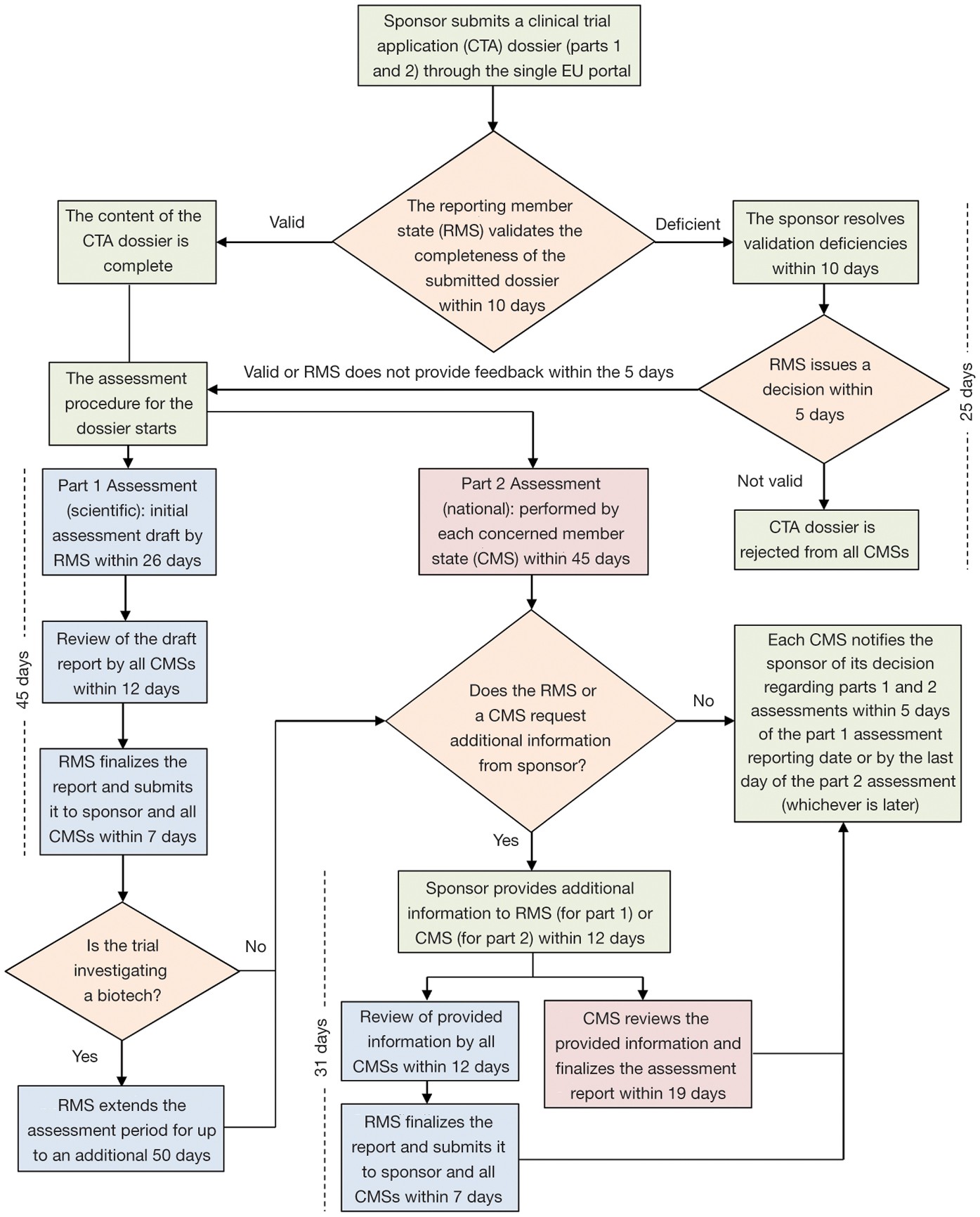
Flow of clinical trials application according to 2001/20/EC Directive.... | Download Scientific Diagram
The Clinical Trials Directive: How Is It Affecting Europe's Noncommercial Research | PLOS Clinical Trials
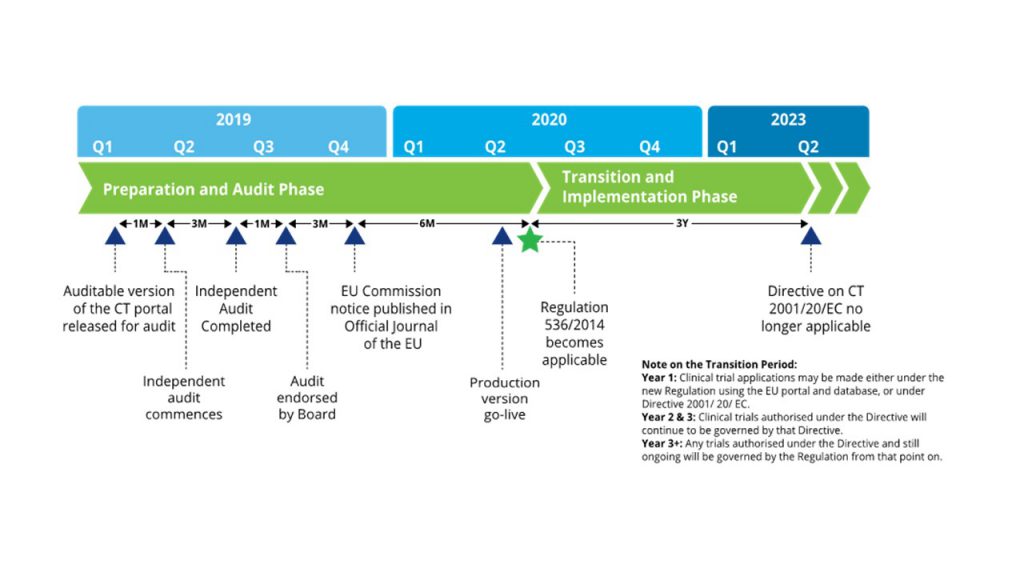
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS