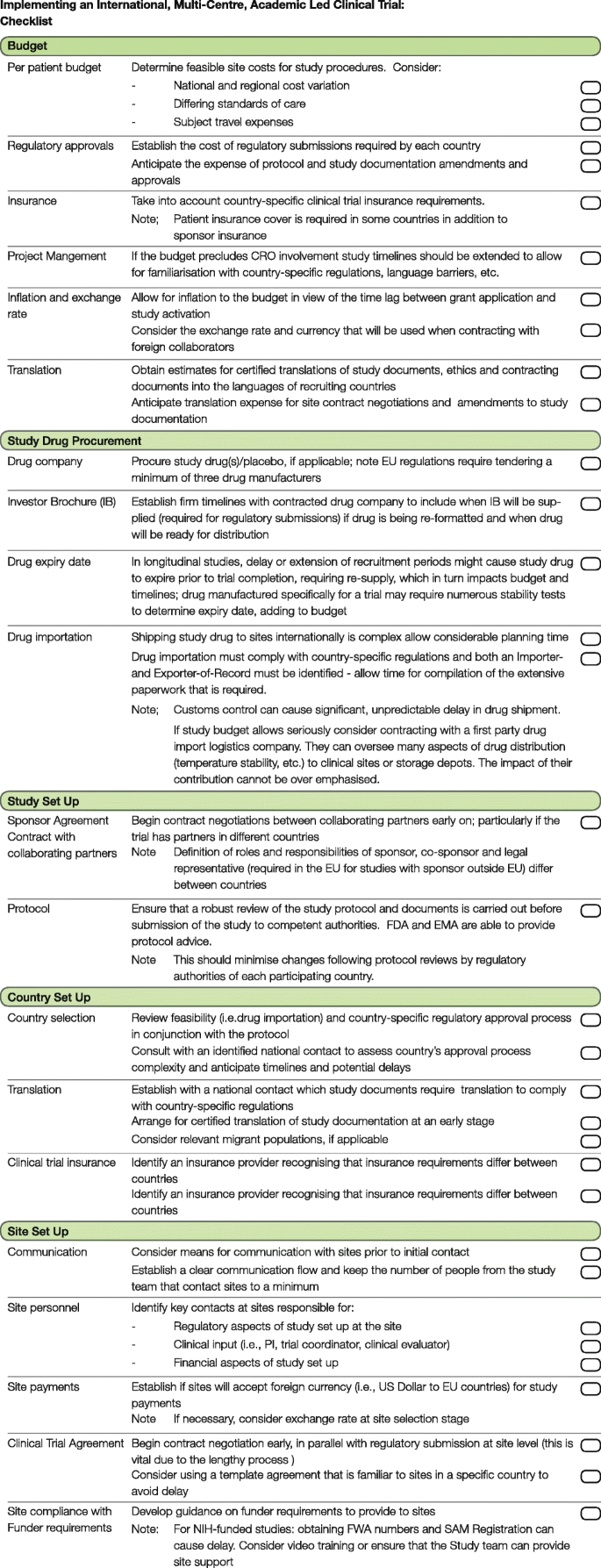
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text
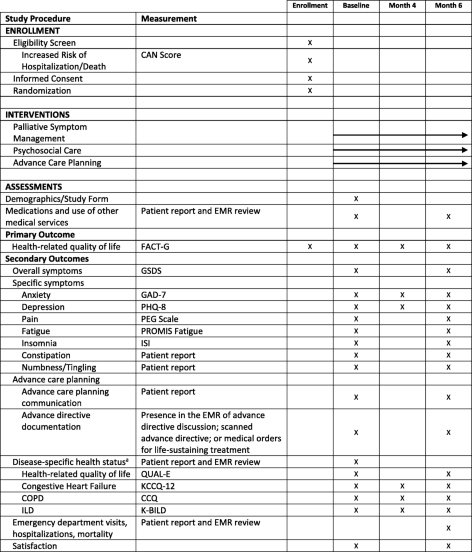
Advancing Symptom Alleviation with Palliative Treatment (ADAPT) trial to improve quality of life: a study protocol for a randomized clinical trial | Trials | Full Text
Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): Extending the CONSORT Statement | PLOS Medicine

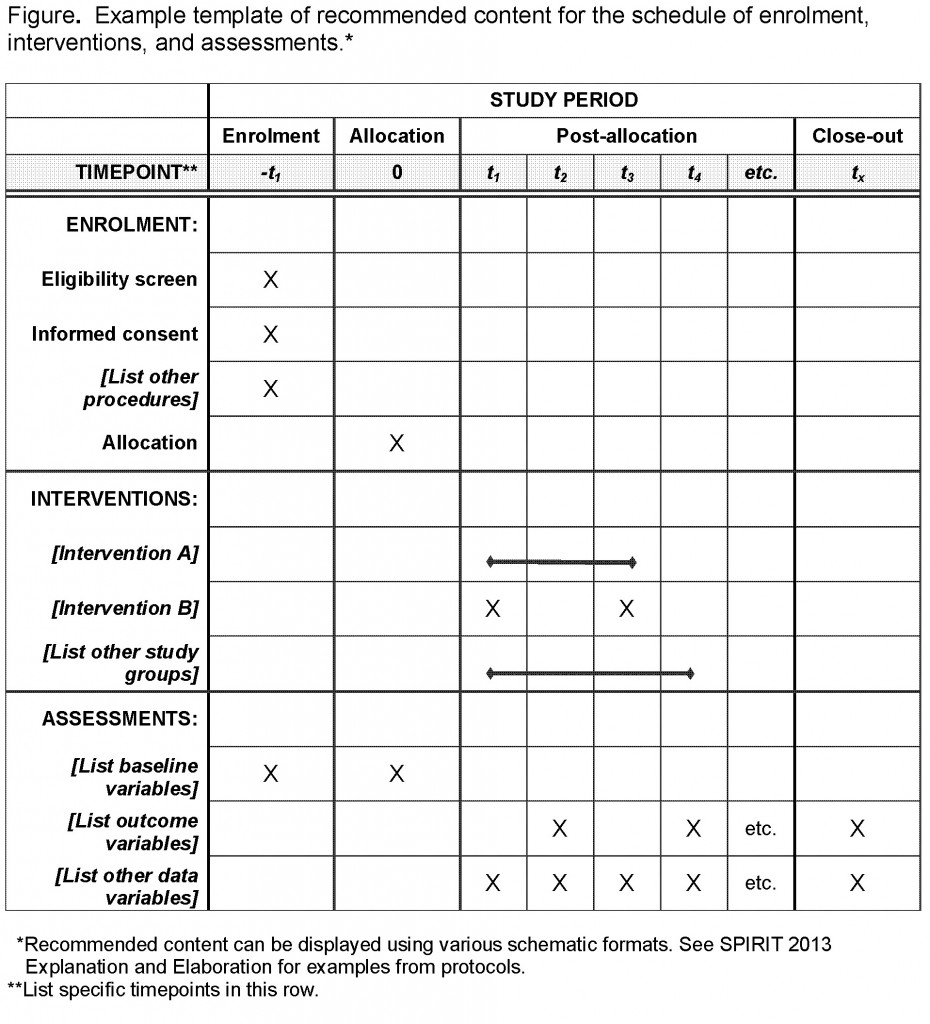
SPIRIT 2013 Checklist: Recommended items to address in a clinical trial... | Download Scientific Diagram
![PDF] SPIRIT 2013 statement: defining standard protocol items for clinical trials. | Semantic Scholar PDF] SPIRIT 2013 statement: defining standard protocol items for clinical trials. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ccf1230c8d03a35229dc9fd36776d69c3356cfa3/6-Table2-1.png)
PDF] SPIRIT 2013 statement: defining standard protocol items for clinical trials. | Semantic Scholar

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Development of a checklist of quality indicators for clinical trials in resource-limited countries: The French National Agency for Research on AIDS and Viral Hepatitis (ANRS) experience | Semantic Scholar
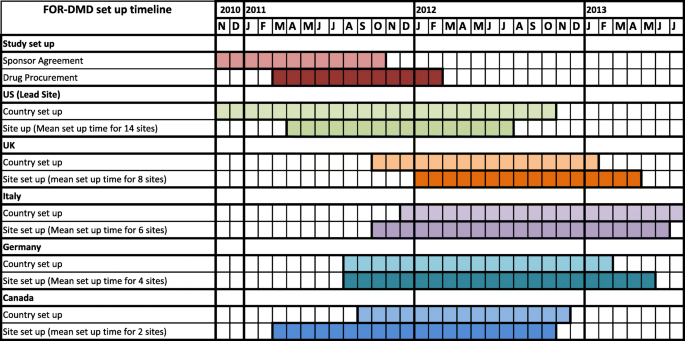
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text

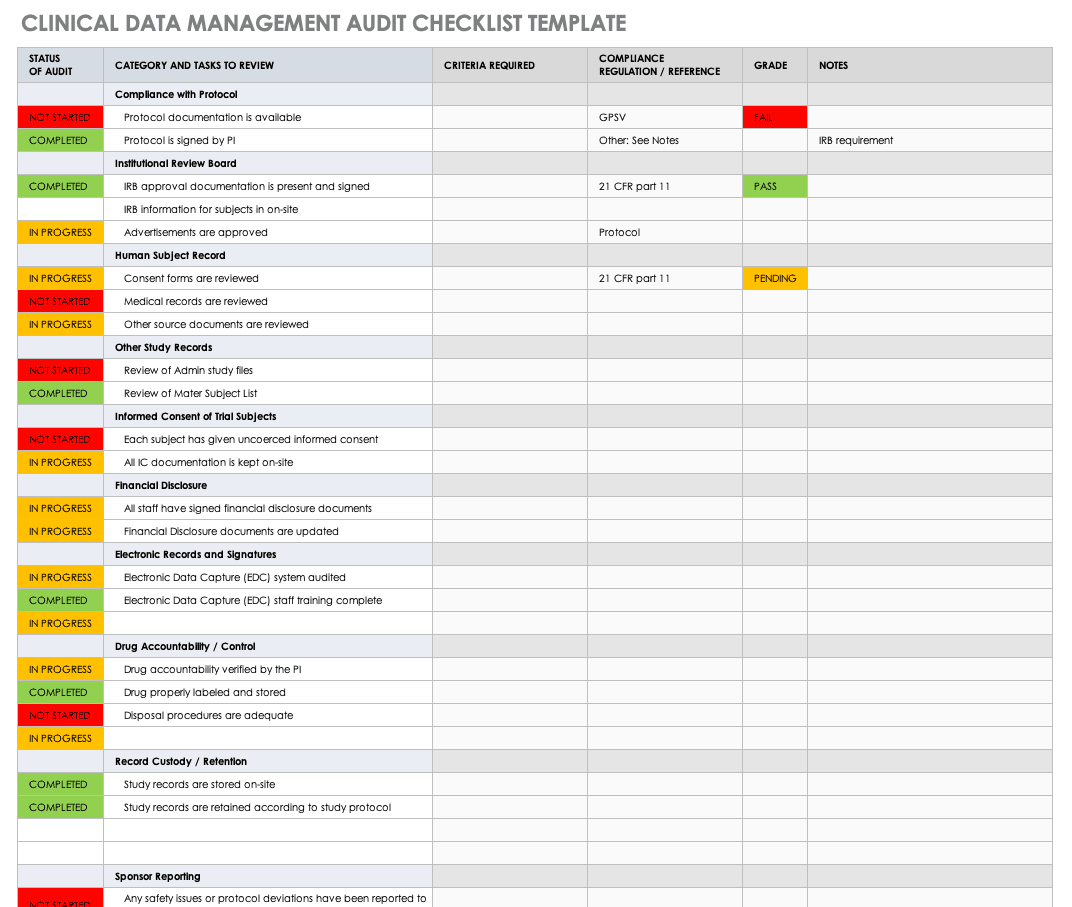
Clinical trials best practice checklist: Guidance for Australian clinical research sites from CT:IQ - ScienceDirect

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - The Lancet Digital Health
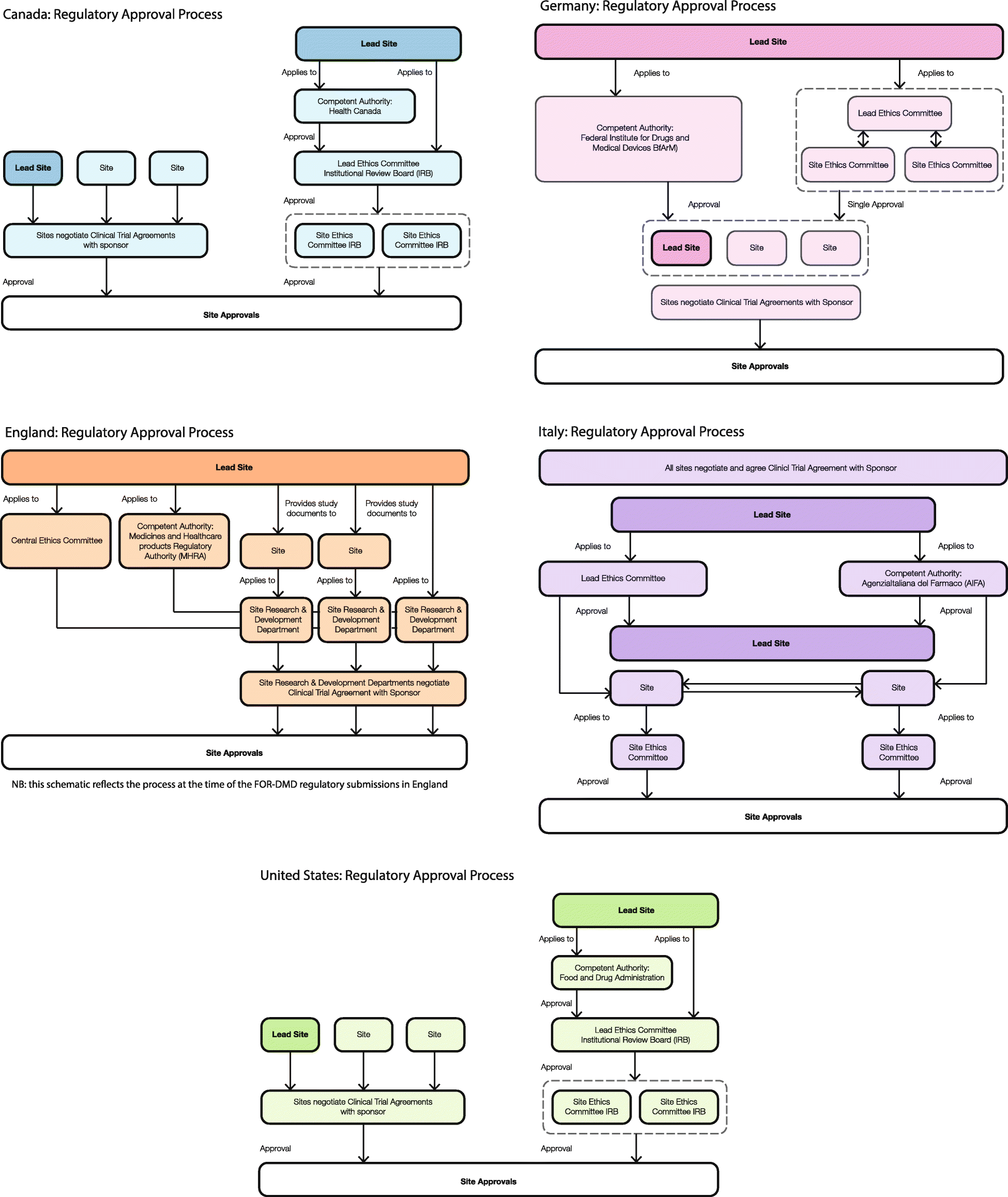
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text

Development of a Good Clinical Practice inspection checklist to assess clinical trial sites in Vietnam | BMJ Open