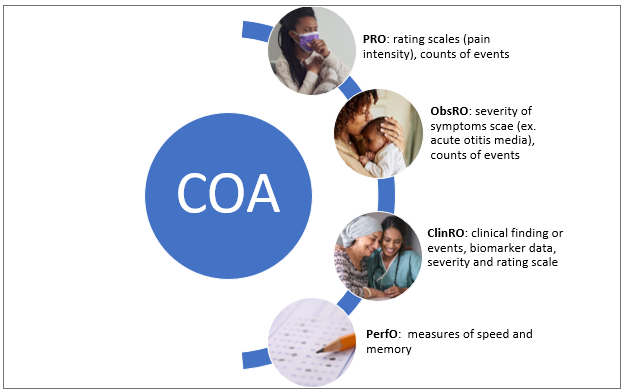
This Paper focuses on CDISC Questionnaires, Ratings and Scales (QRS) supplements and types of FDA Clinical Outcome Assessments
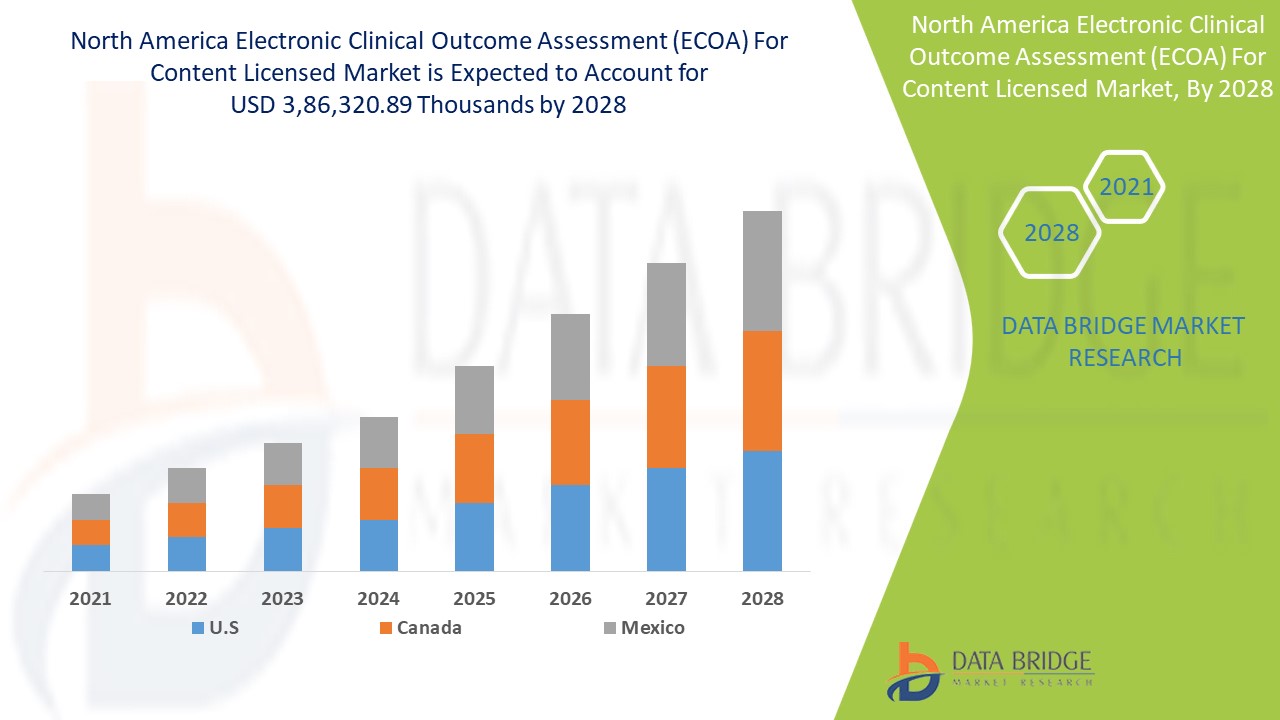
North America Electronic Clinical Outcome Assessment (eCOA) for Content Licensed Market Report – Industry Trends and Forecast to 2028 | Data Bridge Market Research
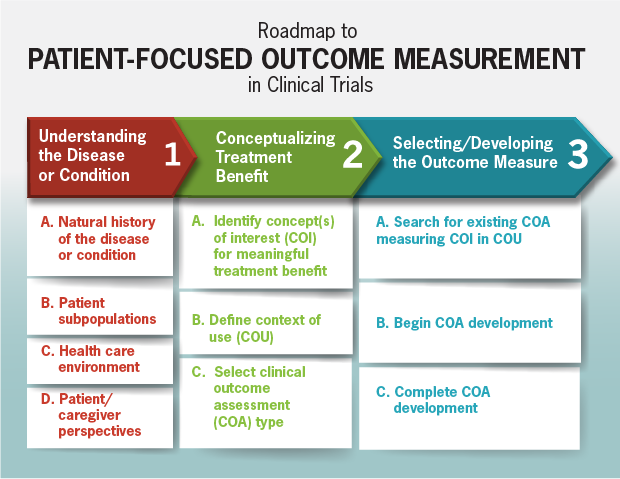
Patient-Focused Drug Development: Selecting, Developing, or Modifying Fit-for-Purpose Clinical Outcome Assessments Snapshot

Clinical Outcome Assessments: Conceptual Foundation—Report of the ISPOR Clinical Outcomes Assessment – Emerging Good Practices for Outcomes Research Task Force - ScienceDirect

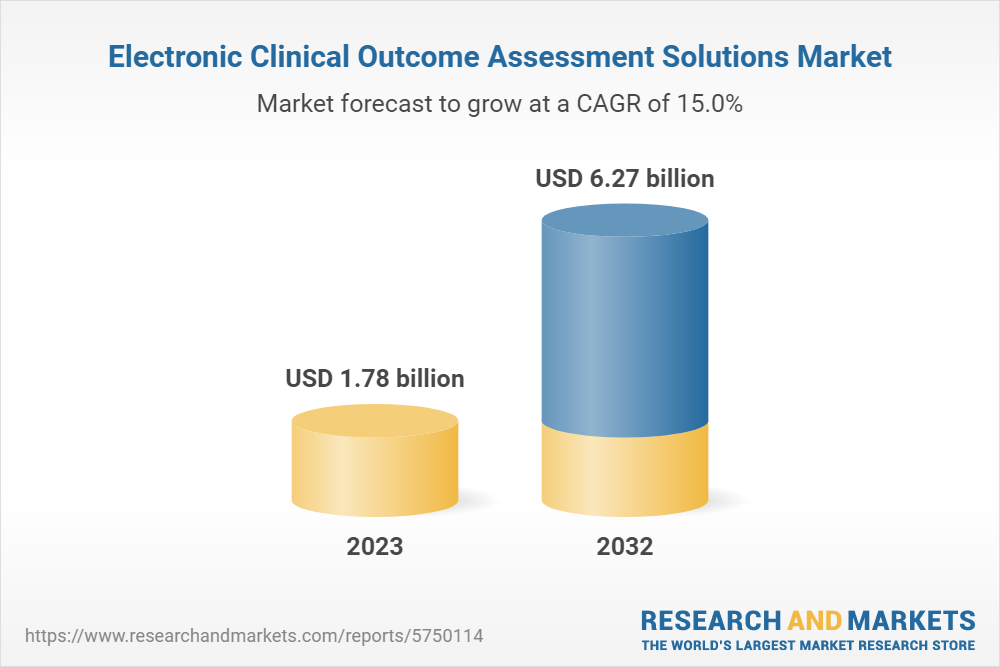
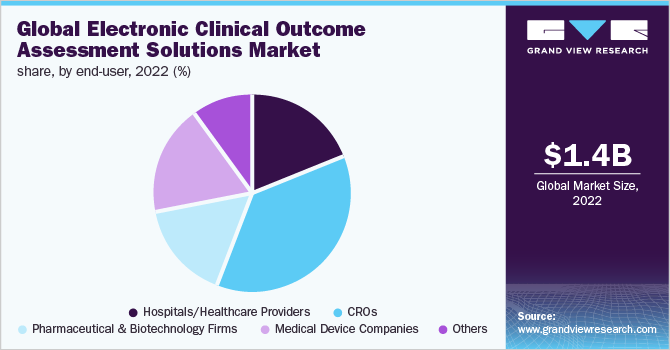
Electronic Clinical Outcome Assessment Solutions Market Overview with Share Analysis, Dynamics, Top Trends, and Regional Analysis to 2032